Prucalopride for chronic constipation is an important topic for millions of people who have spent years — sometimes decades — trying fiber, MiraLax, senna, and magnesium without lasting relief.
If nothing seems to work long-term, the issue may not be the stool — it may be how your gut moves.
For those who haven’t found success with traditional laxatives, prucalopride offers a different approach. It’s an FDA-approved prescription medication that works on the nerves and muscles of the colon — helping your gut create the natural wave-like movements needed to push stool forward.
Instead of just softening stool, it focuses on restoring how the colon actually moves. For people with slow gut motility, this can make a real difference.
A 40-Year Laxative Cycle — Why This Matters
Imagine dealing with constipation for 40 years. You have tried every fiber supplement, every laxative brand, every “miracle” fruit, every mineral. They work for a few days—then you’re back where you started. Every doctor visit ends the same way: “Eat more fiber. Drink more water.” And yet nothing works for more than a few days.
This experience is not rare. Chronic constipation affects an estimated 9%–20% of the global population. In the US alone, roughly 35 million adults live with Chronic Idiopathic Constipation — and for many of them, the root cause has nothing to do with how much fiber they eat. Research shows that CIC patients use an average of 3 OTC products before seeking help from a healthcare provider, and only 6% ever use prescription treatments.
The turning point for the person whose experience inspired this article came from a Naturopathic Doctor who asked a different question: “Is your colon actually moving the way it should?” That question led to a prescription for prucalopride — not a laxative, but a prokinetic agent that targets the neuromuscular system of the gut itself. For the first time in four decades, a provider had addressed the engine of digestion rather than just the stool.
If you have spent years trying OTC solutions without lasting relief, understanding why those solutions may be incomplete — and what the clinical alternatives are — is the goal of this guide. Start with our evidence comparison of OTC laxatives if you want context on what you may have already tried.
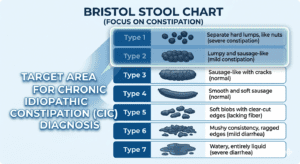
What Is Chronic Idiopathic Constipation? (With Real Data)
Chronic Idiopathic Constipation (CIC) is a functional gastrointestinal disorder defined by persistent, difficult, infrequent, or incomplete bowel movements with no identifiable structural or organic cause. “Idiopathic” means the cause is unknown.
| Statistic | What It Means | Clinical Significance |
|---|---|---|
| 35M+ | US adults estimated to have CIC | CIC affects ~14% of the US adult population — more common than asthma or diabetes |
| <3 | Complete spontaneous bowel movements per week | The clinical threshold for constipation diagnosis; CIC also requires symptoms in ≥25% of defecations |
| 2+ | Failed laxative trials required | Prucalopride is a second-line prescription treatment — at least two laxatives must have been tried first |
| $6,284 | Average annual outpatient cost per CIC patient | CIC patients incur significantly higher healthcare costs than those without the condition |
CIC Is More Than Discomfort — It Has Real Life Impact
Research consistently shows that CIC is not a minor inconvenience. A study using the National Health and Wellness Survey found that CIC patients reported significantly lower quality of life scores than matched controls, alongside substantially higher levels of work impairment:
| Measure | CIC Patients | Matched Controls |
|---|---|---|
| Absenteeism (missed work) | 9.08% | 5.20% |
| Presenteeism (reduced productivity at work) | 29.52% | 19.09% |
| Overall work impairment | 33.65% | 21.56% |
| Daily activity impairment | 46.58% | 33.90% |
| Provider visits (past 6 months) | 7.73 | 5.63 |
Source: Sun et al., Digestive Disease Sciences (2011) — National Health and Wellness Survey, n=1,430 CIC patients matched to controls.
Studies also confirm that the health burden of CIC can be higher than that of Type 2 diabetes, IBS, and GERD in some quality-of-life measures. This matters because it reinforces why stepping up from failed OTC treatment to an evidence-backed prescription option is clinically justified — not an overreaction.
For patients who also deal with motility problems related to the gut microbiome, our article on gut microbiome rebalancing for constipation explains the connection between dysbiosis and serotonin signaling.
Why Fiber and Laxatives Often Fall Short for CIC
Standard first-line advice — “eat more fiber, drink more water” — works well for occasional, dietary-related constipation. For CIC with impaired gut motility, the same advice repeatedly misses the root cause. Here is why each common option has limitations for this patient group:
Osmotic Laxatives (MiraLax / PEG 3350)
Polyethylene glycol draws water into the colon to soften stool. It is among the best-evidenced OTC options — the AGA–ACG gives it a strong recommendation for CIC. However, PEG does not address why the colon is failing to propel waste forward. Long-term daily use can create reliance on external osmotic assistance rather than restoring natural motility. See our complete OTC laxative guide for a full breakdown.
Stimulant Laxatives (Senna, Bisacodyl / Dulcolax)
Stimulants irritate the intestinal lining to trigger a contraction. Effective for acute episodes, but the AGA–ACG gave stimulants only a conditional recommendation for CIC — and guidelines warn that prolonged daily use can reduce colonic response over time. They are not intended for the kind of daily long-term use that many CIC patients resort to.
Bulk-Forming Fiber (Psyllium / Metamucil)
Psyllium has the strongest evidence base among fiber supplements and received a conditional recommendation from the AGA–ACG. However, head-to-head studies show it is less effective than PEG for stool frequency. For slow-transit CIC, adding bulk to a stalled system can actually worsen bloating and discomfort without resolving the underlying propulsion problem. Our guide to sugar-free fiber supplements for seniors covers safer options for older adults with dietary restrictions.
Research shows that in some patients, the gut’s serotonin signaling system — which tells the colon when to contract — doesn’t work properly. When this signal is disrupted, the colon slows down and struggles to move waste forward.
👉 That’s why adding more fiber or water often doesn’t solve the problem.
If the colon isn’t moving effectively, extra bulk can simply sit there — leading to more bloating and discomfort.
A simple way to think about it:
it’s like pressing the accelerator when the engine itself isn’t working properly.
What Is Prucalopride for Chronic Constipation? (Motegrity Explained)
Prucalopride is a selective, high-affinity serotonin type 4 (5-HT4) receptor agonist — a prokinetic agent FDA-approved on December 14, 2018, for the treatment of Chronic Idiopathic Constipation in adults. It is marketed in the US as Motegrity by Takeda Pharmaceuticals.
| Feature | Detail |
|---|---|
| Drug class | Selective 5-HT4 receptor agonist (prokinetic) |
| FDA approval date | December 14, 2018 |
| US brand name | Motegrity (Takeda Pharmaceuticals) |
| Generic available? | Yes — ANI Pharmaceuticals, launched January 2025 |
| Available doses | 1 mg and 2 mg film-coated tablets |
| How taken | Once daily, with or without food |
| Standard adult dose | 2 mg/day (1 mg/day for patients ≥65 or severe renal impairment) |
| AGA–ACG guideline rating | Strong recommendation — highest level |
| Prescription required? | Yes — second-line after ≥2 failed laxative trials |
The joint AGA–ACG Clinical Practice Guideline on CIC pharmacological management gave prucalopride a strong recommendation based on high-quality evidence — the same level as PEG, linaclotide, and plecanatide, and a higher level than fiber, senna, lactulose, or magnesium oxide (all conditional only).
How Prucalopride Works: The Gut-Serotonin Pathway, Explained
Most people associate serotonin with mood and the brain. What is less widely known is that over 95% of the body’s total serotonin is produced in the gut — by enterochromaffin (EC) cells lining the intestinal mucosa. This gut-derived serotonin is the primary chemical trigger for peristalsis: the coordinated, wave-like muscle contractions that move food and waste through the colon.
In many patients, this signaling pathway is compromised, leading to slow-transit issues. This is precisely where Prucalopride for chronic constipation acts by targeting the 5-HT4 receptors directly on the myenteric neurons. By binding to these receptors, the drug stimulates the release of neurotransmitters that restore natural, wave-like movement to a stalled system.
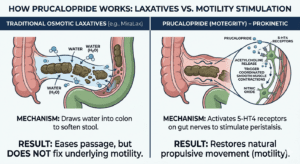
Comparison: Natural Signaling vs. Prucalopride Action
| Step | Healthy Colon (Natural Process) | CIC Patient with Prucalopride |
| Trigger | Meal / Food intake | Prucalopride Tablet |
| Chemical | Natural Serotonin (5-HT) released | Drug binds to 5-HT4 receptors |
| Action | 5-HT4 Receptors activated naturally | Bypasses serotonin deficiency |
| Response | Myenteric Neurons fire | Myenteric Neurons fire (restored) |
| Result | Normal Peristaltic Contraction | Restored Colonic Motility |
Prucalopride works by targeting specific serotonin (5-HT4) receptors in the colon that control bowel movement. By activating these receptors, it helps restore natural muscle contractions in the gut.
What makes it different from older drugs is its high selectivity — meaning it mainly works on the gut receptors and has very little effect on the heart.
Older medications in the same class, like cisapride and tegaserod, were removed from the market because they were less selective and could affect heart rhythm. Prucalopride was designed to avoid that issue, making it a safer option in its class.
2. Neurotransmitter Cascade
Receptor activation triggers release of acetylcholine, calcitonin gene-related peptide (CGRP), and nitric oxide from myenteric neurons. Acetylcholine causes upstream smooth muscle to contract; nitric oxide causes downstream smooth muscle to relax. This is the classic, coordinated peristaltic wave.
3.Enhanced, Coordinated Peristalsis
Unlike stimulant laxatives that force a sudden, uncoordinated “flush,” prucalopride works with the gut’s existing neuromuscular architecture. The result is augmented but physiologically normal propulsive contractions — the colon moves waste forward the way it was designed to.
4.Normalized Colonic Transit Time
The goal is to restore normal colonic transit time — the rate at which waste moves through the large intestine. For slow-transit CIC patients, this translates to more frequent, complete, and comfortable bowel movements. This is why onset can occur within 1–2 days, not weeks.
The gut microbiome also plays a role in this serotonin pathway. Research in Scientific Reports shows that gut dysbiosis can upregulate the serotonin transporter (SERT), reducing available 5-HT and further impairing colonic motility. This is why supporting the microbiome alongside prokinetic therapy may offer complementary benefit — explored further in our article on probiotics and chronic constipation.
Clinical Trial Results for Prucalopride for Chronic Constipation
Prucalopride’s FDA approval rests on six randomized, double-blind, placebo-controlled trials involving 2,484 adult CIC patients across the US, Europe, and Asia-Pacific. Five lasted 12 weeks; one was a 24-week Phase 4 trial.
| Trial Outcome | Prucalopride | Placebo |
|---|---|---|
| ≥3 CSBMs/week (primary endpoint) | 27.8% | 13.2% |
| Clinically meaningful improvement in bowel function | 47% | 29.9% |
| Primary endpoint met (of 6 trials) | 5 of 6 | — |
| Odds ratio (OR) for ≥3 CSBMs/week | OR 2.68 | p<0.001 |
| Time to first improvement | As early as Week 1 | — |
| Discontinuation due to side effects | 5% | 3% |
Key Findings Clinicians Cite
- Onset confirmed at 1–2 days in the 2025 Cureus narrative review — faster than most patients expect
- 4 mg is NOT more effective than 2 mg: a published meta-analysis confirmed the 4 mg dose provided no additional benefit over 2 mg (OR 0.96 vs. placebo — not statistically significant). This is a critical clinical point often overlooked.
- Works equally in men and women, and in patients aged under and over 65
- Significant reduction in rescue laxative use in the prucalopride arm (p < 0.001)
- Benefits sustained across both 12-week and 24-week study durations
Still Using OTC Laxatives Without Lasting Relief?
Understand what the evidence actually says about each OTC option — and when the research suggests it may be time to discuss prescription alternatives with your doctor.
How it compares to common laxatives (and why that matters)
| Feature | Prucalopride (Motegrity) | MiraLax (PEG) | Senna/Bisacodyl | Psyllium Fiber |
|---|---|---|---|---|
| Drug Class | 5-HT4 agonist (prokinetic) | Osmotic laxative | Stimulant laxative | Bulk-forming fiber |
| Primary Action | Stimulates natural colonic peristalsis via nerve signaling | Draws water into colon to soften stool | Irritates gut lining to force contraction | Adds bulk and water content to stool |
| Best For | Chronic motility-based CIC after ≥2 laxatives have failed | Occasional to short-term relief | Acute, short-term only | Mild, dietary-related constipation |
| Addresses Root Cause? | ✔ Yes — colonic motility | ✗ No | ✗ No | ✗ No |
| AGA–ACG Guideline Level | ✔ Strong recommendation | ✔ Strong recommendation | Conditional only | Conditional only |
| Long-Term Use Safety | ✔ Designed for ongoing use | Safe short-term; reliance risk long-term | ✗ Reduced response over time | ✔ Generally safe |
| Prescription Required? | Yes (Rx only) | No (OTC) | No (OTC) | No (OTC) |
| Onset of Action | 1–2 days | 12–72 hours | 6–12 hours | 1–3 days |
| Cardiac Safety | ✔ Confirmed (no MACE increase) | ✔ Safe | Monitor long-term | ✔ Safe |
| Most Common Side Effects | Headache, nausea (transient, first 1–2 days) | Bloating, cramping | Cramping, urgency, dependency risk | Bloating, gas (initially) |
| Approximate Cost | Brand: expensive; Generic: more affordable | ~$10–25/month OTC | ~$5–15/month OTC | ~$15–30/month OTC |
Side Effects, Warnings & Contraindications
Most side effects are mild and usually appear in the first 1–2 days after starting the medication. For many people, they go away quickly as the body adjusts.
The most commonly reported effects include headache, nausea, mild stomach discomfort, or changes in bowel habits like looser stools at the beginning.
In clinical studies, very few people stopped the medication because of side effects — most were able to continue treatment without major issues.

Common Adverse Reactions (≥2% incidence in trials)
| Reaction | Frequency/Timing | Typical Severity |
| 🤕 Headache | Most common; first 1–2 days | Mild to Moderate |
| 🤢 Nausea | Early treatment phase | Transient (goes away quickly) |
| 😣 Abdominal Pain | Occasional cramping | Usually resolves in 1 week |
| 💨 Diarrhea | Initial response to motility | Mild and short-lived |
| 🌀 Dizziness | Reported in clinical trials | Rare/Transient |
| 😴 Fatigue | First few doses | Mild |
| 🤮 Vomiting | Less frequent | Resolves after adaptation |
| 💨 Flatulence | Common initially | Decreases over time |
The 2025 Cureus narrative review notes headache onset is typically within 2 days and resolves without intervention in most patients. Nausea and diarrhea also typically resolve within the first week of treatment without requiring discontinuation.
Contraindications — Do NOT Use Prucalopride If You Have:
- Known hypersensitivity to prucalopride (reactions: dyspnea, rash, pruritus, urticaria, facial edema)
- Intestinal perforation or obstruction due to structural or functional gut wall disorder
- Obstructive ileus
- Severe inflammatory bowel disease: Crohn’s disease, ulcerative colitis, toxic megacolon / megarectum
- Severe renal impairment (CrCl <30 mL/min) — dose must be reduced to 1 mg/day; not eliminated
Dosing — Quick Reference
| Patient Group | Starting Dose | Notes |
|---|---|---|
| Adults under 65 | 2 mg once daily | Standard dose; taken with or without food |
| Adults 65 and older | 1 mg once daily | Escalate to 2 mg if insufficient response; 81% of elderly patients in trials were escalated |
| Severe renal impairment (CrCl <30) | 1 mg once daily | Do not escalate beyond 1 mg |
| Moderate/mild renal impairment | 2 mg once daily | No dose adjustment required |
| Hepatic impairment (any level) | No adjustment | Prucalopride not significantly metabolized by the liver |
Note: Prucalopride is a CYP 3A4 substrate. Always disclose all concurrent medications to your prescriber. Insufficient safety data exists for pregnancy and breastfeeding — consult your physician.
Who Qualifies for Prucalopride for Chronic Constipation?
The AGA–ACG guideline positions prucalopride as a second-line prescription option for adults with CIC who have not achieved adequate relief from at least two conventional laxative classes. The following clinical profile describes a likely candidate:
- Chronic constipation (≤2–3 CSBMs/week) persisting for at least 6 months
- Hard stools, straining, or incomplete evacuation in ≥25% of defecations
- Inadequate response to at least two OTC laxative trials (e.g., fiber AND osmotic/stimulant)
- No identifiable underlying cause: not IBS, Crohn’s, hypothyroidism, or drug-induced
- Constipation significantly impacts quality of life, work, social activities, or mental health
- No history of intestinal obstruction, perforation, or severe IBD
- Adequate renal function (or willing to use adjusted 1 mg dose if CrCl <30)
- Cardiac history cleared by prescribing physician
Supportive OTC Products — Honest Reviews
Many CIC patients use OTC supplements alongside prokinetic therapy — or while awaiting a prescription evaluation — to support general gut health. Below are four evidence-informed options with fully honest assessments including limitations. These are supportive aids, not substitutes for proper medical treatment of CIC.
Best Microbiome Support
Garden of Life Dr. Formulated Probiotics — Once Daily
50 Billion CFU | 16 strains incl. B. lactis, L. rhamnosus GG | Shelf-stable | NSF Certified Gluten-Free | Non-GMO
Research published in Scientific Reports demonstrates that gut dysbiosis can upregulate SERT (serotonin transporter), suppressing 5-HT availability in the colon and slowing transit — the same pathway prucalopride targets. Supporting the microbiome with clinically studied strains — particularly B. lactis HN019 and L. rhamnosus GG, both present in this formula — may offer modest complementary benefit. A 2017 meta-analysis of 21 probiotic trials found these strains can modestly improve stool frequency and reduce colonic transit time after 4–8 weeks. Formulated by Dr. David Perlmutter, a board-certified neurologist and microbiome specialist. Available in women’s (with L. reuteri, L. fermentum for vaginal health) and men’s (with added colon/immune support) formulas.
✅ Pros
- Multiple clinically studied strains
- Shelf-stable — no refrigeration
- Third-party potency verified
- Includes organic prebiotic fiber blend
- Men’s and women’s formulas available
❌ Cons
- 4–8 weeks before bowel effects
- Histamine-producing strains in women’s formula
- Large capsule size
- Premium price ($35–45/month)
View on Amazon [Affiliate link — we earn a small commission at no extra cost to you.]
Best Osmotic Support
Natural Vitality CALM — Magnesium Citrate Powder
Magnesium citrate | Multiple flavors | ~325 mg magnesium per serving | No artificial sweeteners in original formula
Magnesium citrate works via a gentle osmotic mechanism — drawing water into the intestines to soften stool — without irritating the gut lining or creating the dependency risk associated with stimulant laxatives. Research confirms its effectiveness for functional constipation, and it is frequently used as a short-term bridge while patients await prescription evaluation or as an adjunct during the early weeks of prokinetic therapy. Magnesium also supports smooth muscle relaxation throughout the body, including the intestinal wall, which may reduce the straining component of constipation.
✅ Pros
- Gentle osmotic action, no irritation
- Well-researched for constipation
- Also supports relaxation and sleep
- Easy to take as a flavored drink
❌ Cons
- Unsafe in kidney disease without guidance
- Loose stools if overdosed
- Multiple drug interactions
- Does not restore gut motility
View on Amazon [Affiliate link — we earn a small commission at no extra cost to you.]
Best Fiber Supplement
Metamucil Psyllium Fiber — Sugar-Free
Psyllium husk soluble fiber | Sugar-free powder | AGA–ACG conditional recommendation | Multiple sizes
Psyllium husk has the strongest clinical evidence base among all OTC fiber supplements. Unlike fermented fibers (inulin, wheat dextrin, guar gum) that lose their gelling properties before reaching the large intestine, psyllium retains its water-holding gel structure throughout the entire GI tract — softening stool and improving its consistency for passage. For CIC patients who additionally have hard, difficult-to-pass stools, psyllium can be a useful stool-consistency adjunct alongside prokinetic therapy. The AGA–ACG gave it a conditional recommendation — evidence-based, but below the strong recommendation given to PEG and prucalopride. For senior patients, see our sugar-free fiber guide for seniors.
✅ Pros
- Best-evidenced OTC fiber supplement
- Improves stool consistency
- Sugar-free option available
- Affordable (~$15–25/month)
❌ Cons
- Choking risk without adequate water
- Initial bloating (1–2 weeks)
- Less effective than PEG for frequency
- Does not improve colonic motility
View on Amazon [Affiliate link — we earn a small commission at no extra cost to you.]
Best OTC Osmotic
MiraLax Laxative Powder — PEG 3350
Polyethylene glycol 3350 | Tasteless, odorless | Dissolves in any liquid | AGA–ACG strong recommendation | Multiple sizes
PEG 3350 (MiraLax) shares the AGA–ACG’s strong recommendation alongside prucalopride — though for its respective OTC role as a first-line and short-term treatment. It is tasteless, dissolves in any liquid, and produces far less cramping and urgency than stimulant laxatives. For CIC patients who are starting prucalopride, physicians sometimes recommend PEG during the initial 2–4 weeks to manage breakthrough symptoms while the prokinetic takes full effect. The large cardiovascular safety study of prucalopride itself used PEG as its comparator group — validating both agents as safe within their appropriate clinical roles. For elderly patients, our elderly constipation relief guide covers how to use PEG safely as a home option.
✅ Pros
- AGA–ACG strong recommendation
- Tasteless and easy to mix
- No cramping or urgency
- Safe short-term for most adults
❌ Cons
- Not a long-term CIC management solution
- Does not restore gut motility
- Dependency risk with prolonged daily use
- Occasional bloating at higher doses
View on Amazon [Affiliate link — we earn a small commission at no extra cost to you.]
Cost & Access Strategies in the US (2026)
Brand-name Motegrity remains expensive without prescription drug coverage. Here are the most practical strategies to reduce cost:
| Strategy | How It Helps | Who It Applies To |
|---|---|---|
| Generic prucalopride (ANI, 2025) | Substantially lower cost than brand-name; ask pharmacist for “prucalopride tablets” | All patients |
| Takeda patient assistance program | May provide Motegrity at low or no cost | Uninsured or underinsured patients |
| Motegrity copay savings card | Reduces monthly out-of-pocket copay | Insured patients |
| GoodRx / RxSaver comparison | Compare generic prices across pharmacies — can vary significantly by location | All patients |
| Insurance prior authorization | Most insurers require documentation of ≥2 failed laxative trials — compile this history with your GI doctor | Patients with insurance |
A note on prior authorization: most insurance plans require documentation that at least two OTC laxative classes were tried without adequate relief. If you have kept records of what you tried, for how long, and what happened, this documentation can significantly speed up the authorization process. Your gastroenterologist can often complete the paperwork on your behalf.
Frequently Asked Questions
Is this just another laxative?
No. Laxatives mainly work by changing the stool — either by softening it, adding water, or increasing bulk to make it easier to pass.
Prucalopride works differently.
Instead of acting on the stool, it helps stimulate the natural muscle movements of your colon so waste can move through more effectively.
This is why it may help some people who haven’t responded well to regular laxatives.
Can I take prucalopride long-term?
Yes. Unlike stimulant laxatives that can reduce colonic sensitivity over time, prucalopride is designed for ongoing use in adults with CIC. Clinical safety data covers up to 24 weeks in controlled trials, with open-label extension data for longer durations. Research on treatment cessation and re-treatment confirms that effects return promptly when treatment is restarted after a break — and do not worsen after stopping. Your physician will monitor your response and adjust as needed.
How quickly does prucalopride work for chronic constipation?
How quickly does Prucalopride for chronic constipation work? Most patients see results within 1–2 days, though it may take up to 4 weeks to see the full clinical benefit of Prucalopride for chronic constipation management.
What happens if I stop taking prucalopride?
Research from re-treatment trials shows that when prucalopride is discontinued, bowel function returns toward baseline — the drug does not worsen long-term colonic function. When treatment is restarted, the response returns promptly. This distinguishes it from stimulant laxatives, which can reduce colonic sensitivity with prolonged use.
Do the AGA and ACG actually recommend prucalopride?
Yes — the joint AGA–ACG Clinical Practice Guideline issued a strong recommendation for prucalopride in adult CIC management, the highest endorsement level, based on high-quality evidence from multiple randomized controlled trials. This places it in the top tier alongside PEG, linaclotide, and plecanatide — and above fiber, senna, lactulose, and magnesium oxide, all of which received only conditional recommendations.
Who should NOT take prucalopride?
Patients with intestinal obstruction, perforation, or severe IBD (Crohn’s, ulcerative colitis, toxic megacolon) must not use this medication. Severe renal impairment (CrCl <30 mL/min) requires dose reduction to 1 mg/day. Insufficient safety data exists for pregnancy and breastfeeding — consult your physician. Always disclose all current medications to your prescriber.
Can it be taken alongside probiotics or fiber supplements?
Generally yes — these target different mechanisms (motility vs. microbiome vs. stool consistency) and there are no known major pharmacological interactions with standard probiotic or fiber supplements. Always confirm with your prescribing physician before adding new supplements, as individual circumstances vary.
Is prucalopride approved for children?
No. As of 2026, prucalopride is FDA-approved only for adult patients. Post-marketing pediatric studies (ages 6 months–18 years) were required by the FDA but have not yet produced a pediatric indication.
|
Medical Disclaimer:
This article is for educational purposes only and not medical advice. Prucalopride (Motegrity) is a prescription drug; consult a doctor before use. Content reviewed for clinical accuracy by Dr. ABM Sadikullah, MBBS, FCPS (Medicine).
Affiliate Disclosure: ConstipationRelief.net may earn a commission from links at no extra cost to you. This does not affect our honest reviews or editorial integrity.
References & Sources
- U.S. Food and Drug Administration. Drug Trial Snapshots: Motegrity (prucalopride). December 2018. fda.gov
- Takeda Pharmaceuticals America. Motegrity (prucalopride succinate) Full Prescribing Information. Updated July 2025. rxdruglabels.com
- Camilleri M, et al. Efficacy and safety of prucalopride in chronic constipation: integrated analysis of 6 RCTs in 2484 patients. Digestive Diseases and Sciences. 2016. PMC4943977
- Ford AC, et al. Efficacy of prucalopride in chronic constipation: systematic review and meta-analysis of RCTs. Clinical Gastroenterology and Hepatology. 2012. cghjournal.org
- Sahu SK, et al. Pharmacotherapy for CIC Beyond Conventional Laxatives: Narrative Review. Cureus. November 2025. PMC12722192
- Lacy BE, et al. AGA–ACG Clinical Practice Guideline: Pharmacological Management of Chronic Idiopathic Constipation. PMC10542656
- Sun S, et al. Impact of chronic constipation on HRQoL, work productivity, and healthcare resource use: National Health and Wellness Survey analysis. Digestive Disease Sciences. 2011. PMID: 21380761
- Nag A, et al. The Humanistic and Economic Burden of CIC in the USA: A Systematic Literature Review. Clinical and Experimental Gastroenterology. 2020. PMC7371558
- Ge X, et al. Dysbiosis contributes to chronic constipation via regulation of serotonin transporter. Scientific Reports. 2017. DOI: 10.1038/s41598-017-10835-8
- Quigley EMM. An update on prucalopride in the treatment of chronic constipation. Therapeutic Advances in Gastroenterology. 2017. PMC5673021
- ANI Pharmaceuticals. FDA Approval and Launch of Prucalopride Tablets with 180-Day CGT Exclusivity. GlobeNewswire. January 2, 2025.
- McRorie JW. Evidence-Based Approach to Fiber Supplements and Clinically Meaningful Health Benefits, Part 2. Nutrition Today. 2015. PMC4415970